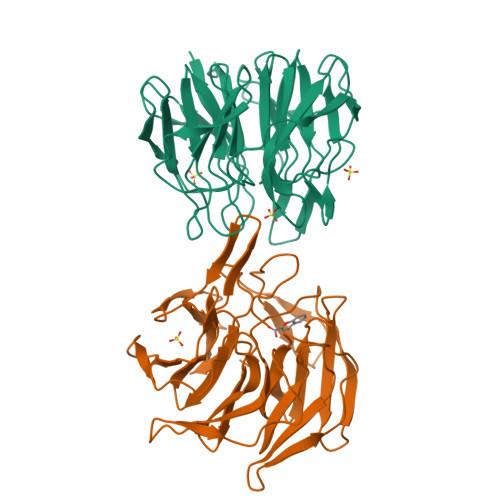
Interaction Energetics and Druggability of the Protein-Protein Interaction between Kelch-like ECH-Associated Protein 1 (KEAP1) and Nuclear Factor Erythroid 2 Like 2 (Nrf2).
Zhong, M., Lynch, A., Muellers, S.N., Jehle, S., Luo, L., Hall, D.R., Iwase, R., Carolan, J.P., Egbert, M., Wakefield, A., Streu, K., Harvey, C.M., Ortet, P.C., Kozakov, D., Vajda, S., Allen, K.N., Whitty, A.(2020) Biochemistry 59: 563-581
- PubMed: 31851823
- DOI: https://doi.org/10.1021/acs.biochem.9b00943
- Primary Citation of Related Structures:
5WFL, 5WFV, 5WG1, 5WHL, 5WHO, 5WIY - PubMed Abstract:
Development of small molecule inhibitors of protein-protein interactions (PPIs) is hampered by our poor understanding of the druggability of PPI target sites. Here, we describe the combined application of alanine-scanning mutagenesis, fragment screening, and FTMap computational hot spot mapping to evaluate the energetics and druggability of the highly charged PPI interface between Kelch-like ECH-associated protein 1 (KEAP1) and nuclear factor erythroid 2 like 2 (Nrf2), an important drug target. FTMap identifies four binding energy hot spots at the active site. Only two of these are exploited by Nrf2, which alanine scanning of both proteins shows to bind primarily through E79 and E82 interacting with KEAP1 residues S363, R380, R415, R483, and S508. We identify fragment hits and obtain X-ray complex structures for three fragments via crystal soaking using a new crystal form of KEAP1. Combining these results provides a comprehensive and quantitative picture of the origins of binding energy at the interface. Our findings additionally reveal non-native interactions that might be exploited in the design of uncharged synthetic ligands to occupy the same site on KEAP1 that has evolved to bind the highly charged DEETGE binding loop of Nrf2. These include π-stacking with KEAP1 Y525 and interactions at an FTMap-identified hot spot deep in the binding site. Finally, we discuss how the complementary information provided by alanine-scanning mutagenesis, fragment screening, and computational hot spot mapping can be integrated to more comprehensively evaluate PPI druggability.
Organizational Affiliation:
Acpharis, Inc. , 160 North Mill Street , Holliston , Massachusetts 01746 , United States.