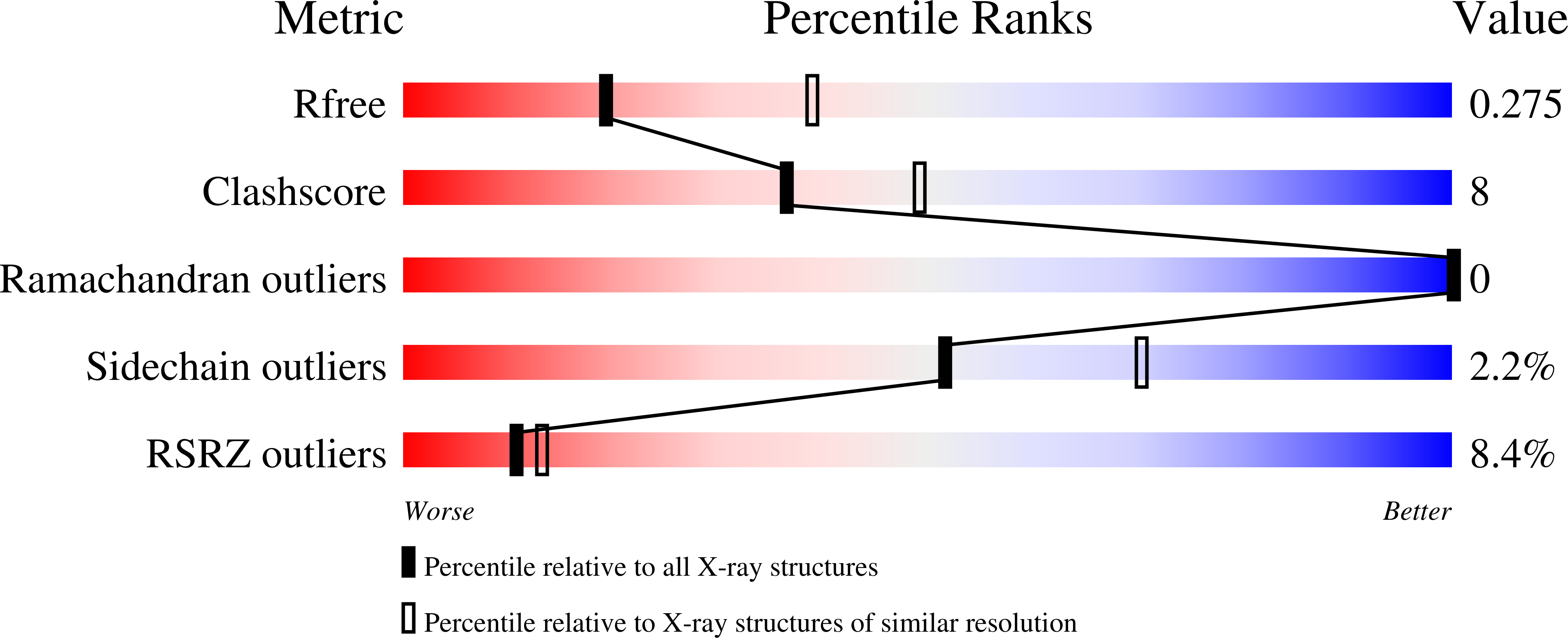
Rational Design of CYP3A4 Inhibitors: A One-Atom Linker Elongation in Ritonavir-Like Compounds Leads to a Marked Improvement in the Binding Strength.
Samuels, E.R., Sevrioukova, I.F.(2021) Int J Mol Sci 22
- PubMed: 33467005
- DOI: https://doi.org/10.3390/ijms22020852
- Primary Citation of Related Structures:
7KVH, 7KVI, 7KVJ, 7KVK, 7KVM, 7KVN, 7KVO, 7KVP, 7KVQ, 7KVS - PubMed Abstract:
Inhibition of the major human drug-metabolizing cytochrome P450 3A4 (CYP3A4) by pharmaceuticals and other xenobiotics could lead to toxicity, drug-drug interactions and other adverse effects, as well as pharmacoenhancement. Despite serious clinical implications, the structural basis and attributes required for the potent inhibition of CYP3A4 remain to be established. We utilized a rational inhibitor design to investigate the structure-activity relationships in the analogues of ritonavir, the most potent CYP3A4 inhibitor in clinical use. This study elucidated the optimal length of the head-group spacer using eleven (series V) analogues with the R 1 /R 2 side-groups as phenyls or R 1 -phenyl/R 2 -indole/naphthalene in various stereo configurations. Spectral, functional and structural characterization of the inhibitory complexes showed that a one-atom head-group linker elongation, from pyridyl-ethyl to pyridyl-propyl, was beneficial and markedly improved K s , IC 50 and thermostability of CYP3A4. In contrast, a two-atom linker extension led to a multi-fold decrease in the binding and inhibitory strength, possibly due to spatial and/or conformational constraints. The lead compound, 3h , was among the best inhibitors designed so far and overall, the strongest binder (K s and IC 50 of 0.007 and 0.090 µM, respectively). 3h was the fourth structurally simpler inhibitor superior to ritonavir, which further demonstrates the power of our approach.
Organizational Affiliation:
Department of Pharmaceutical Sciences, University of California, Irvine, CA 92697-3900, USA.